Cannabis Alchemy
by D.Gold
Isomerization
The oil produced by alcohol extraction and purification with petroleum ether contains tetrahydrocannabinol, two other compounds closely related to THC but nonpsychoactive (cannabidiol and cannabinol), and several other compounds which contribute the taste and smell of the oil. The quality and quantity of the THC in the oil is determined by the quality and potency of the starting material. The oil from very strong cannabis material contains a much higher percentage of THC than the oil from marijuana or hashish that is less potent. The quality of the THC and the characteristics of the effect (high) are determined by the relative positions of the double bonding in the THC molecule. The higher-rotating forms are more potent than the low-rotating and produce a higher, more psychedelic and spiritual effect. Methods for converting THC from low-to-highrotating follow.
The quantity of cannabidiol in the oil is important, as it may be converted to THC, thereby increasing the potency of the oil proportionally. Experience has indicated that the quantity of cannabidiol is usually at least equal to the quantity of THC. Because of this, the strength of the oil can be at least doubled through isomerization, and in some cases potency may be increased five to six times.
By using the correct chemicals and methods to convert the cannabidiol to THC, it is possible simultaneously to convert the THC (that which occurs naturally in the oil and also that which has been produced from cannabidiol) to higher-rotating forms.
The highest benefit is obtained by starting with material high in cannabidiol, isomerizing the cannabidiol to THC, and converting the THC to its higher-rotating form. Both the potency of the oil and the quality of the high are greatly increased. The operation is carried out as follows: The oil from the ether extraction is dissolved in absolute ethanol or pure methanol in the ratio of one gram of oil to ten grams of solvent. The ethanol may be denatured, but must not contain water. One drop of 100 per cent sulfuric acid is added to the alcohol/oil solution for each gram of oil. The acid is added slowly with continuous stirring. Pure sulfuric acid is very strong and will cause severe burns. Safety glasses, long rubber gloves, and clothing that covers as much of the body surface as possible are advised when working with it. Sulfuric acid burns are treated by immediate washing with water and bicarbonate of soda. The sulfuric acid is kept in a safety bottle made by permanently fitting a glass bottle with a screw top in a styrofoam-lined metal can.
A Pyrex pot containing the oil/alcohol/sulfuric acid solution is placed in the refluxing apparatus originally used for refluxing the material in alcohol. Pyrex is substituted for the stainless-steel pot because of the reactive nature of the sulfuric acid. The rig is placed in the boiling water bath and refluxed for two hours. At the end of this time, the stew pot is placed in an ice-water bath and opened. The solution is poured into an equal amount of water and extracted with petroleum ether, as was done in removing the ether soluble oil from the alcohol extract solution. The ether solution is then poured into four volumes of water and gently inverted twenty-five times, releasing the pressure each time. The layers are allowed to separate, the ether/oil layer is siphoned off and the water is discarded. The ether/oil solution is poured into four volumes of 5% bicarbonate of soda solution in water. It is mixed, then separated, and the ether/oil layer is siphoned off. The bicarbonate of soda solution is discarded and the previous step (washing with pure water) is repeated twice. The ether is evaporated from the ether/oil solution, as was done previously in the first purification, using the stew pot apparatus. The pure ether is collected in the pan held in the colander. The oil now contains a much higher percentage of THC (determined by the amount of cannabidiol originally present). The THC is of the high rotating isomeric form, and all of the toxins have been removed from the oil.
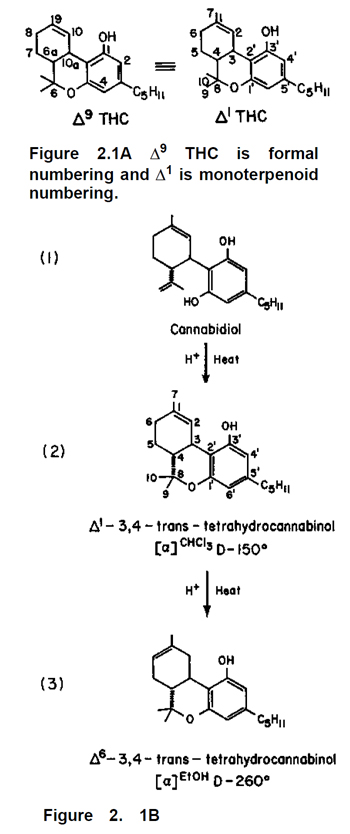
Many readers especially those with some exposure to chemistry, may be curious about what happens to the molecules during the isomerization. The structural formulas for the reaction are given above (Fig. 2 .1B). For the non-organic chemist, this is an opportunity to overcome your fear of formulas. The first credits for finding this reaction go to Roger Adams, who first described it in 1940; a detailed account appeared in 1941 (Adams et al., 1941), but the correct formulas were not discovered until the 1960s. The changes that go on are minor; the first reaction forms a third ring by attaching the oxygen at 1’ to the carbon at 8. The second reaction is just a shifting of one double bond from the 1,2 position to the 6,1 position. Two conventions for numbering the ring system are used in the literature; sometimes Δ1 is called Δ9 and Δ6 is called Δ8 (Mechoulam, 1970). See Figure 2.1A. Since two different THCs are products of the reaction, the question becomes: how much of each is produced? This depends on the reaction conditions. If more concentrated or stronger acid is used and the time of reaction is increased, more Δ6 THC is produced; there is a 90% conversion of Δ1→Δ6 THC if p-toluenesulfonic acid in toluene is used for 10 hours at 100°C (Hively et al., 1966). With mild conditions (absolute ethanol, 0.05% hydrochloric acid boiled for 2 hours) the product is mainly Δ1 THC (Gaoni and Mechoulam, 1964). So, mild conditions give Δ1 THC; more vigorous conditions give Δ6 THC. This sounds simple enough; in reality, however, other factors can play a significant role. Starks (1977) mentions some side reactions, which have been reported; these vary with the solvent and conditions used for the isomerization. There is recent evidence (Bonuccelli, 1979) that solvents such as chloroform which promote free-radical reactions can result in large decreases in THC content. A chloroform solution of marijuana exposed to sunlight for 30 minutes lost 25—35% of its THC; the reaction was slower in the dark but still significant. The authors recommend ethanol over chloroform, and cool, dark storage. Slight decomposition even occurs with ethanol. Given that Δ1 and Δ6 THC are both possible products, another important question is whether there is any difference in the pharmacological effects between the two. Mechoulam (1970) says workers have found them to be about equal in activity, but some differences have been reported. Most studies are carried out on laboratory animals, so the effects measured are not mainly psychological. Psychological effects can be subtle, and the possibility of a difference between the effects of Δ1 and Δ6 THC are yet to be researched. The methodology for measuring such subtle changes remains a challenge to research psychologists.